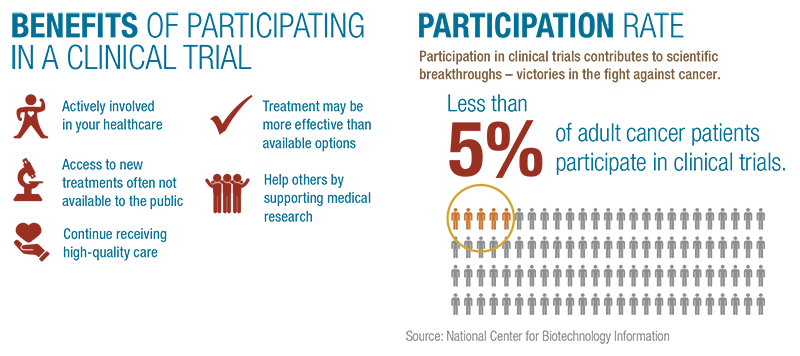
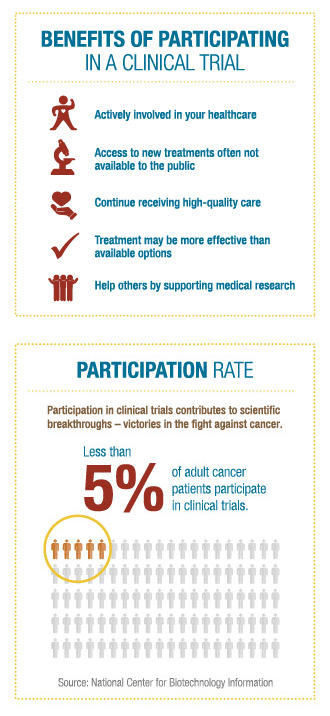
How to Participate
Participating in a clinical trial is an easy, straightforward process. To enroll yourself or your loved one, follow these simple steps:
- Research your options
Texas Oncology is actively involved in a wide range of clinical trials. Information is updated frequently.
- Complete a recruitment screening
Once you’ve identified a trial, discuss your options with your physician to determine if the trial is appropriate for you. You may be asked additional screening questions to ensure the study is a good fit.
- Submit your consent to participate
It’s critical that you discuss the clinical trial with the researcher to understand the risks, benefits, and structure associated with the study. Once the details of the trial are disclosed and your questions have been answered, you will sign a consent agreement.
- Participate in the study
Physicians guide you through the study. Your experience will vary based on the specific type and phase of the trial.
- Provide additional information and samples.
Once you begin participating in the trial, you may need to provide additional information and biological samples (blood, tissue, or genetic tests) to the researchers.
- Review the results.
Many clinical trials openly communicate the trial results with participants. These results often help shape future medical choices and improve the quality of life for cancer patients.